 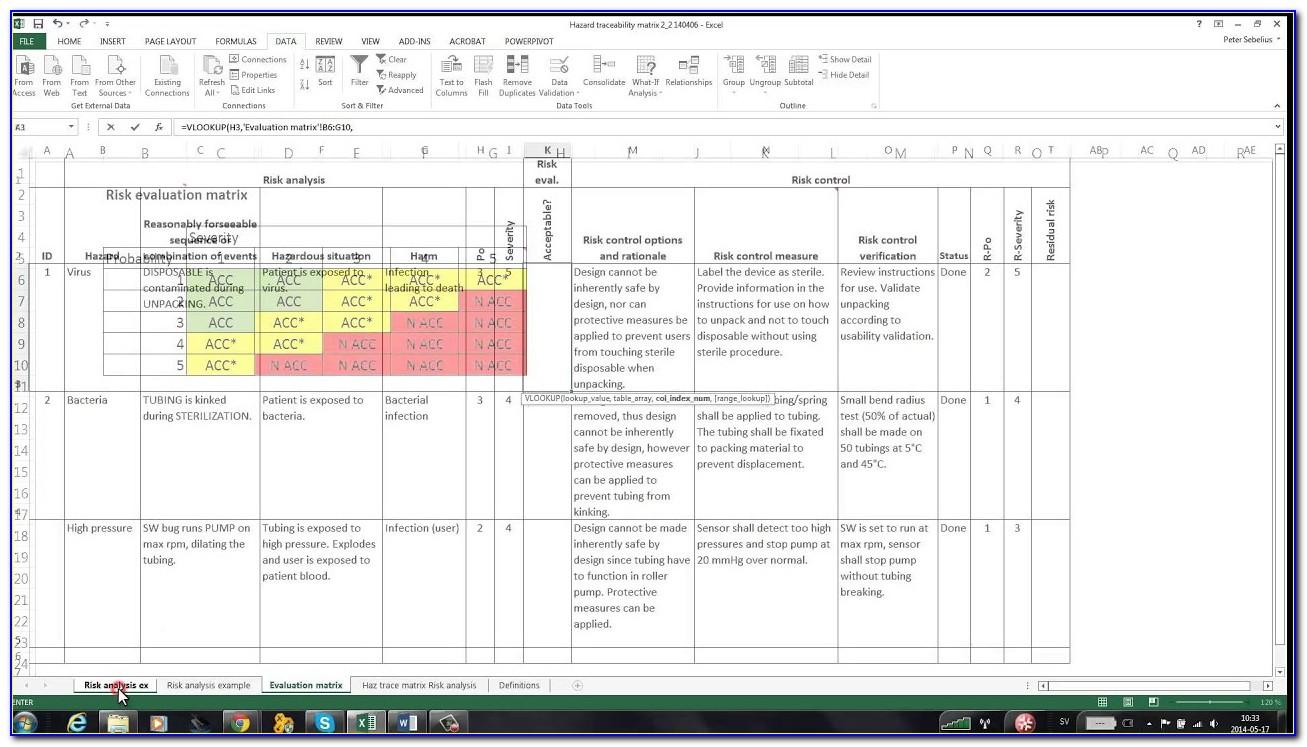
Generally speaking, within the medical device industry, risk analysis consists of recognizing and addressing any factor that might hinder the ability to get your product to market.  
If you start risk analysis in the middle of product development, you are taking an approach that goes against the very definition of the term. Anything less than managing risk throughout the entire product lifecycle is to knowingly risk the health of your end users. You have to begin risk analysis at the very start of the product design phase, and manage it through the manufacturing and distribution phases. 
Conduct Risk Analysis From Beginning to End To be effective, medical device risk analysis must be conducted from start to finish you need to take a holistic, connected approach and it’s important to adopt the right tools. In the medical device industry, managing and mitigating risk can be a challenge, but it is essential because the consequences of doing a sub-par job comes at great cost: time that’s better spent on developing a high quality product, money you didn’t plan on spending, and a reputation that takes years to build can be irreparably damaged within minutes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
